Issue
18
Page
27
Species
|
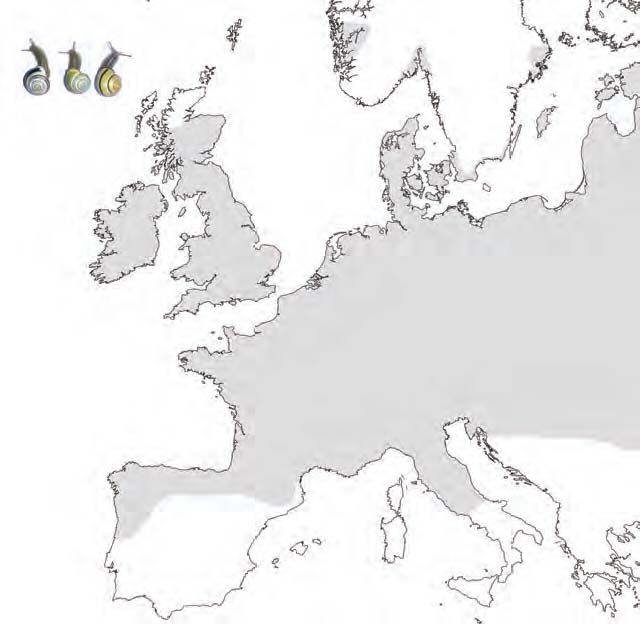
The land snail Cepaea nemoralis is highly polymorphic for shell colour and banding (Figure 1). Within and between populations individuals can display a shell colour of several shades of yellow, pink, or brown, and any number of bands from 0 to 5. Generally C. nemoralis populations are monomorphic for lip colour, with the common morph being dark brown or black. However, in a few European areas white or palelipped individuals are also present in some populations i.e. Cantabria in Spain; the Pyrenees; Denmark; Yorkshire and Cornwall in England; North Wales; Western Scotland; and the west coast of Ireland (Figure 2). These polymorphic populations consist of both the dark-lipped and white-lipped individuals, and additionally pale brown-lipped individuals, which are possibly heterozygotes (Cain et al., 1968). Very rarely, some populations in these areas are monomorphic for the rare lip colour i.e. in the Pyrenees.
The genes for shell colour, lip colour, and banding are very tightly linked forming a supergene (Jones et al. 1977). Yet, while around 98% of British populations are polymorphic for shell colour and banding, lip colour is almost completely invariant, with there being only a few known British locations that contain populations that are polymorphic for this trait. It is therefore unusual that while natural selection operates so that shell colour and banding are variable, the monomorphism in the lip colour is probably maintained by strong stabilising selection.
There are several linked hypotheses that may explain the distribution of white-lipped C. nemoralis: 1) repeated evolution of the same character due to common environment or selection 2) the populations are derived from the same Pleistocene refugial populations or 3) introgression of the white-lip allele from the sister species Cepaea hortensis. The aim of the project was to establish by means of genetic methods as to whether the populations containing individuals with the rare lip colour all have a common origin. Additionally, the project would also contribute towards understanding the postglacial colonisation of Britain and Ireland.
The last glacial period
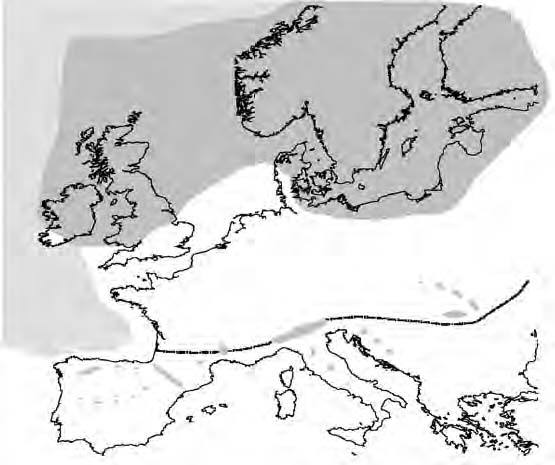
The Last Glacial Maximum (LGM; 23 to 18 kyr ago) was at its height around 18 kyr ago during the Pleistocene, when the European ice sheet extended as far south as 52°N with an area of permafrost stretching to 47°N (Hewitt, 2004)(Figure 3). Glaciers also formed on the Southern European Mountains, such as the Alps and Pyrenees, creating a barrier that to a great extent blocked the pathways of migrating species. It is therefore assumed that the majority of temperate species, such as Cepaea nemoralis, survived within the ice-free southern peninsulas i.e. Iberia, Italy, and the Balkans (Bennett 1997; Hewitt, 1999).
The end of the Younger Dryas period (~ 10 Kyr ago) marked the beginning of the latest interglacial phase, the Holocene. Species that were previously confined to refugial areas tracked the warming climate, generally migrating in a northward direction, and filled any available niches en route (Hewitt, 1999). Perhaps modern day European populations that are polymorphic for lip colour all originated from the same southern refugial area, most likely Iberia.
Field Work
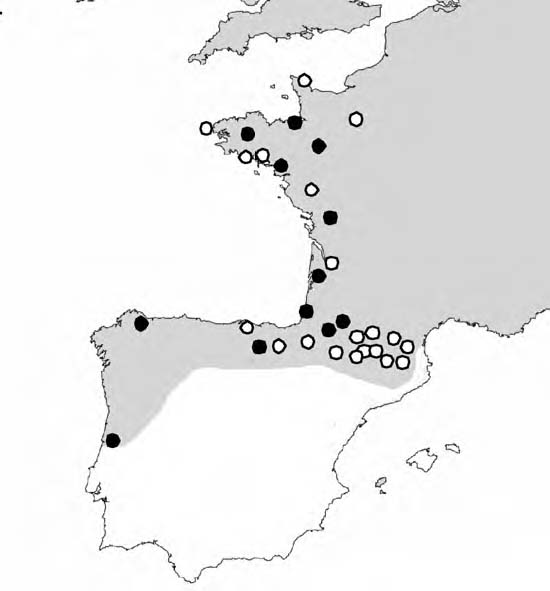
During the summer of 2007, I conducted a transect down the west coast of France, then across the Pyrenees and the north coast of Spain. In all, I collected over 200 individuals from 20 separate locations (Figure 4). Populations were located by thorough inspection of suitable habitats, with care taken so as not to sample near sites where recent introductions are likely, such as, near agriculture areas, parks, or private gardens. Where possible between 10 and 30 individuals were collected at each site, from an area no larger than 10 x 10m. Samples were returned to the lab and frozen on arrival.
The research so far
To date, fragments of the mtDNA gene cytochrome oxidase subunit I (COI) and 16S rRNA for over 950 Cepaea nemoralis individuals from >100 Western European sites have been sequenced and analysed. Intriguingly, the data strongly suggests that individuals from the west coast of Ireland are derived from populations in the Pyrenees, supporting the long-known “Lusitanian” origin of the Irish fauna. Some populations from both of these areas also contain a high proportion of large, white-lipped C. nemoralis. However, there is little evidence that other white-lipped populations in mainland Britain and Europe are primarily derived from the Iberian populations. Additionally, there is no evidence for introgression from the sister species Cepaea hortensis.
Future work
The next step for this study is to compare the Irish Cepaea nemoralis mitochondrial DNA patterns to the fossil records from both the east and west coast of Ireland (Preece et al., 1986; Speller, 2006). Additionally, palaeoclimatic niche modeling of refugial areas in Iberia may be constructed to establish the potential distribution and movement of Spanish C. nemoralis during the Pleistocene and Holocene. Finally, sequencing of additional genes, such as a noncoding nuclear gene, and microsatellite work would be useful to support original findings.
Acknowledgements
I am extremely grateful to the Conchological Society for helping to fund my field work last summer. I would like to thank my supervisor, Dr. Angus Davison, for his help and advice on the project, and the University of Nottingham for funding the lab work. In addition, many thanks to Prof. Robert Cameron and Prof. Steve Jones for their useful discussions about the project.
References
Bennett, K. 1997 Evolution and ecology: the pace of life. Cambridge University Press. Cain, A.J., Sheppard, P.M., and King, J.M.B. 1968 The Genetics of Some Morphs and Varieties of Cepaea nemoralis (L.), Phil. Trans. Roy. Soc. B. 253, 383–396.
Hewitt, G. 1999 Postglacial recolonization of European Biota. Biol. J. Linn. Soc. 68, 87–112.
Hewitt, G. 2004 Genetic consequences of climatic oscillations in the Quaternary. Phil. Trans. R. Soc. Lond. B. 359, 183–195. Jones, J. S., Leith, B. H., and Rawlings, P. 1977 Polymorphism in Cepaea: A problem with too many solutions? Ann. Rev. Ecol. Syst. 8, 109-143.
Preece, R. C., Coxon, P., and Robinson, J. E. 1986 New biostratigraphic evidence of the Post-glacial colonization of Ireland and for Mesolithic forest disturbance Journal of Biogeography 13, 487-509.
Speller, G. R. Molluscan biostratigraphy, stable isotope analyses and dating of Irish Holocene tufas University of Cambridge; 2006.
Thomaz, D., Guiller, A., and Clarke, B. 1996 Extreme divergence of mitochondrial DNA within species of pulmonate land snails Proc. R. Soc. Lond. B 263, 363-368.
|
Figure 1. Diagram to show a selection of the shell pattern polymorphisms of Cepaea nemoralis. A. Dark pink, unbanded. B. Bright pink, unbanded. C. Pale pink, unbanded. D. Dark yellow, unbanded. e. Dark yellow, 5-banded (12345). F. Dark yellow, 5-banded (spread bands). G. Faint pink, with fused bands. H. Dark Yellow, with fused bands. I. Dark yellow, 3-banded (00345). J. Yellow-white, 5-banded (12345). K. Pale yellow, 5-banded (12345). L. Pale yellow, punctate bands. M. Pale yellow, faint bands. N. Dark yellow, mid-banded (00300). o . Pink, mid-banded (00300).
Figure 2. Map to show areas in which populations containing white-lipped individuals are found (circled). The approximate distribution of Cepaea nemoralis is indicated by shading. Insert shows the lip colour polymorphism of C. nemoralis (left: white-lip; middle: pale brown-lip; right: dark-lip).
Figure 3. Map to show the extent of the ice sheet and permafrost in Europe during the Last Glacial Maximum (~18,000 years ago). The grey area represents the ice sheet; sea ice is indicated by the hatched area; the dotted line shows the extent of the permafrost (adapted from Hewitt, 1999).
Figure 4. Map to show the field trip collection sites (white circles; black circles represent sampling sites from Thomaz et al, 1996). The distribution of Cepaea nemoralis is indicated by shading.
|